4.0 Clinical Particulars
4.1 Therapeutic Indications
Zorvin KID Syrup are indicated for treatment of bronchospasm in patients above 6 years with reversible obstructive airway disease.
4.2 Posology and Method of Administration
Route of administration: oral
- Children (Aged 6 to 11 years): 5 ml (1mg) three times daily.
- Adolescents and Adults (Above 12 years of age): 5-10ml (1 to 2mg) thrice daily.
4.3 Contraindications
Zorvin KID Syrup are contraindicated
- In patients with a history of hypersensitivity to any of their components.
- It should not be used for threatened abortion during the first or second trimester of pregnancy.
- Levosalbutamol and beta-blocking drugs such as propranolol should not usually be prescribed together.
4.4 Special Warnings and Precautions for use
General
Paradoxical Bronchospasm
If paradoxical bronchospasm occurs, levosalbutamol should be discontinued immediately and alternative therapy instituted. It should be recognized that paradoxical bronchospasm, when associated with inhaled formulations, frequently occurs with the first use.
Deterioration of Asthma
Asthma may deteriorate acutely over a period of hours or chronically over several days or longer. If the patient needs more doses of levosalbutamol than usual, this may be a marker of destabilization of asthma and requires re-evaluation of the patient and treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g. corticosteroids.
Use of Anti-Inflammatory Agents
Levosalbutamol is not a substitute for corticosteroids. The use of beta-adrenergic agonist alone may not be adequate to control asthma in many patients. Early consideration should be given to adding anti-inflammatory agents, e.g. corticosteroids, to the therapeutic regimen.
Cardiovascular Effects
Levosalbutamol like other beta-adrenergic agonists, can produce clinically significant cardiovascular effects in some patients, as measured by heart rate, blood pressure, and symptoms. Although such effects are uncommon after administration of levosalbutamol at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the t-wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Therefore, levosalbutamol, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
Do Not Exceed Recommended Dose
Do not exceed the recommended dose. Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs in patients with asthma. The exact cause of death is unknown, but cardiac arrest following an unexpected development of a severe acute asthmatic crisis and subsequent hypoxia is suspected.
Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of levosalbutamol or racemic salbutamol. Reactions have included urticaria, angio-oedema, rash, bronchospasm, anaphylaxis, and oropharyngeal oedema. The potential for hypersensitivity must be considered in the clinical evaluation of patients who experience immediate hypersensitivity reactions while receiving levosalbutamol.
Coexisting Conditions
Levosalbutamol, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, hypertension, and cardiac arrhythmias; in patients with convulsive disorders, hyperthyroidism, or diabetes mellitus; and in patients who are unusually responsive to sympathomimetic amines. Clinically significant changes in systolic and diastolic blood pressure have been seen in individual patients and could be expected to occur in some patients after the use of any beta-adrenergic bronchodilator. Changes in blood glucose may occur. Large doses of intravenous racemic salbutamol have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis.
Hypokalaemia
As with other beta-adrenergic agonist medications, levosalbutamol may produce significant hypokalaemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring supplementation.
4.5 Drug Interactions
Short-Acting Bronchodilators
Avoid concomitant use of other short-acting sympathomimetic bronchodilators or epinephrine in patients being treated with levosalbutamol. If additional adrenergic drugs are to be administered by any route, they should be used with caution to avoid deleterious cardiovascular effects.
Beta-blockers
Beta-adrenergic receptor blocking agents not only block the pulmonary effect of beta-adrenergic agonists such as levosalbutamol, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g. prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers should be considered, although they should be administered with caution.
Diuretics
The ECG changes or hypokalaemia that may result from the administration of non-potassium-sparing diuretics (such as loop and thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the co-administration of beta-agonists with non-potassium-sparing diuretics. Consider monitoring potassium levels.
Digoxin
Mean decreases of 16% and 22% in serum digoxin levels were demonstrated after single-dose intravenous and oral administration of racemic salbutamol, respectively, to normal volunteers who had received digoxin for 10 days. The clinical significance of these findings for patients with obstructive airway disease who are receiving levosalbutamol and digoxin on a chronic basis is unclear. Nevertheless, it would be prudent to carefully evaluate the serum digoxin levels in patients who are currently receiving digoxin and levosalbutamol.
Monoamine Oxidase Inhibitors or Tricyclic Antidepressants
Levosalbutamol should be administered with extreme caution to patients being treated with monoamine oxidase (MAO) inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents, because the action of levosalbutamol on the vascular system may be potentiated. Consider alternative therapy in patients taking MAO inhibitors or tricyclic antidepressants.
Halogenated anaesthetics
Owing to the additional antihypertensive effect, there is increased uterine inertia with risk of haemorrhage; in addition, serious ventricular rhythm disorders due to increased cardiac reactivity, have been reported on interaction with halogenated anaesthetics. Treatment should be discontinued, whenever possible, at least 6 hours before any scheduled anaesthesia with halogenated anaesthetics.
4.6 Use in Special Population
Pregnancy
Use of oral levosalbutamol in pregnant or nursing mothers should be considered only if the expected benefit to the mother is greater than any possible risk to the foetus or the infant.
Lactation
It is not known whether levosalbutamol is excreted in human milk. Caution should be exercised when oral levosalbutamol is administered to a nursing mother.
Pediatric Use
Safety and effectiveness of levosalbutamol in pediatric patients below the age of 6 years have not been established.
Renal Impairment
Salbutamol is known to be substantially excreted by the kidney, and the risk of toxic reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
4.7 Effects on Ability to Drive and Use Machines
Zorvin KID syrup is not expected to have any effect on the ability to drive and use machines.
4.8 Undesirable Effects
Potentially serious hypokalaemia may result from beta2-agonist therapy. This effect may be potentiated by hypoxia. Caution is advised in severe asthma; in such cases, monitoring of serum potassium levels is recommended.
Other common side effects such as palpitation, fine tremors of the skeletal muscle (particularly the hand), and muscle cramps may occur.
The other likely side effects are gastrointestinal disturbances such as nausea, vomiting, burning substernal or epigastric pain, and diarrhea. In some cases, nervousness, headache, dizziness, fatigue, and sleeplessness may occur.
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
4.9 Overdose
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the side effects, e.g. tachycardia, nervousness, headache, tremor, nausea, dizziness, fatigue, and sleeplessness. Hypokalaemia may also occur. As with all sympathomimetic medications, cardiac arrest and even death may be associated with the abuse of levosalbutamol. Treatment consists of discontinuation of levosalbutamol together with appropriate symptomatic therapy. The judicious use of a cardioselective beta-receptor blocker may be considered, bearing in mind that such medication can produce bronchospasm. There is insufficient evidence to determine if dialysis is beneficial for overdosage of levosalbutamol.
5.0 Pharmacological Properties
5.1 Mechanism of Action
Levosalbutamol is a selective beta-2 adrenoceptor agonist. At therapeutic doses it acts on the beta-2 adrenoceptors of bronchial muscle providing short acting (4 to 6 hour) bronchodilation in reversible airways obstruction.
Activation of beta2-adrenergic receptors on airway smooth muscle leads to the activation of adenyl cyclase and to an increase in the intracellular concentration of cyclic-3′, 5′-adenosine monophosphate (cyclic AMP). The increase in cyclic AMP is associated with the activation of protein kinase A, which, in turn, inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in muscle relaxation.
5.2 Pharmacodynamic Properties
Levosalbutamol relaxes the smooth muscles of all airways, from the trachea to the terminal bronchioles. Increased cyclic AMP concentrations are also associated with the inhibition of the release of mediators from mast cells in the airways. Levosalbutamol acts as a functional antagonist that relaxes the airway irrespective of the spasmogen involved, thereby protecting against all bronchoconstrictor challenges.
Data indicate that there are beta-receptors in the human heart, 10–50% of which are beta2-adrenergic receptors. All beta-adrenergic agonist drugs can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or ECG changes.
5.3 Pharmacokinetic Properties
Absorption
Both enantiomers of salbutamol are well absorbed from the gastrointestinal tract, with tmax of 45–360 minutes. (S)-salbutamol has a longer tmax when given alone than in the racemate, likely due to (R)-salbutamol–induced changes in gastrointestinal motility. The bioavailability of (S)-salbutamol is about 70% after single and steady-state doses, while (R)-salbutamol increases from 9% (single dose) to 30% at steady state.
Distribution
The blood-to-plasma ratio of salbutamol is about 0.96 ± 0.13, indicating that blood and plasma clearance are similar at steady state. Minimal binding to blood components and similar volumes of distribution suggest that protein binding has little influence on salbutamol enantiomer disposition.
Metabolism
(R)-Salbutamol is metabolized up to 12 times more efficiently than (S)-salbutamol, with significant inter-individual variation in human tissues. SULT1A3 expression is highest in the intestine, indicating that the intestine is the main site of enantioselective presystemic metabolism of orally absorbed salbutamol.
Elimination
(S)-Salbutamol is almost always found in higher amounts in urine than (R)-salbutamol, regardless of the route of administration.
6.0 Nonclinical Properties
6.1 Animal Toxicology or Pharmacology
Not available
7.0 Description
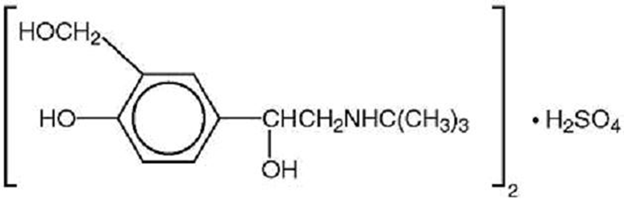
Zorvin-KID Syrup contains levosalbutamol sulfate, a relatively selective beta 2-adrenergic bronchodilator.
Levosalbutamol Sulfate structure
Molecular Formula: (C13H21NO3)2•H2SO4
Molecular Weight: 576.7 g/mol
8. Pharmaceutical particulars
8.1 Incompatibilities
None.
8.2 Shelf-Life
Refer on pack.
8.3 Packaging Information
100 ml bottle
8.4 Storage and Handling Instructions
Store below 25°C. Protect from light. Keep out of reach of children.
9.0 Patient Counselling Information
- The action of Zorvin-KID syrup may last up to 6 hours or longer. Zorvin-KID syrup should not be taken more frequently than recommended.
- Do not increase the dose or frequency of Zorvin-KID syrup without consulting your physician.
- If you find that treatment with Zorvin-KID syrup becomes less effective for symptomatic relief, your symptoms get worse, and/or you need to take the product more frequently than usual, you should seek medical attention immediately. While you are taking Zorvin-KID syrup, other asthma medications and inhaled drugs should be taken only as directed by your physician.
- Common adverse effects include palpitations, chest pain, rapid heart rate, and tremor or nervousness. If you are pregnant or nursing, contact your physician about use of Zorvin-KID syrup.
- Effective and safe use of Zorvin-KID syrup includes an understanding of the way that it should be administered.
10.0 Date of Revision
16th March 2026

