4.0 Clinical Particulars
4.1 Therapeutic Indications
For the symptomatic relief of bronchospasm in bronchial asthma & chronic bronchitis.
4.2 Posology and Method of Administration
Route of administration: oral
| Children between 2 to 6 years | 2.5 ml three times daily |
|---|
| Children between 6 to 12 years | 5 ml three times daily |
|---|
| Adults and children above 12 years | 10 ml three times daily |
|---|
Zorvin-AG syrup should not be used with other cough and cold medicines. It is recommended to take Zorvin-AG syrup with food.
4.3 Contraindications
- Hypersensitivity to any of the components of the formulation.
- It should not be used in patients with preexisting ischemic heart disease or those patients with significant risk factors for ischemic heart disease.
- Thyrotoxicosis.
4.4 Special Warnings and Precautions for use
Levosalbutamol
Levosalbutamol, like other sympathomimetic amines, should be used cautiously in patients with cardiovascular disorders (e.g., ischemic heart disease, hypertension, arrhythmia), hyperthyroidism, diabetes, hypersensitivity to sympathomimetics, or seizure disorders. It may cause significant changes in blood pressure and should be used with caution in patients with severe heart disease; patients should seek medical advice if they experience chest pain or worsening cardiac symptoms. Symptoms such as dyspnoea or chest pain should be carefully evaluated as they may be cardiac or respiratory in origin. Due to its positive inotropic effect, it should be avoided in hypertrophic cardiomyopathy. Potentially serious hypokalaemia may result from ß2-agonist therapy. Caution is advised in acute severe asthma as this effect may be potentiated by hypoxia and by concomitant treatment with xanthine derivatives, steroids and diuretics. Serum potassium levels should be monitored in such situations.
Ambroxol Hydrochloride
Ambroxol should be used with caution in patients with gastric ulceration and in those with impaired mucus production or clearance. Avoid contact with eyes and skin, as well as ingestion or inhalation. In conditions like malignant cilia syndrome, the benefits of mucus liquefaction should be weighed against the risk of secretion retention. Adequate fluid intake enhances its secretolytic effect. Concomitant use with antitussives should be avoided due to the risk of mucus obstruction. Very rare cases of severe skin reactions (e.g., Stevens–Johnson syndrome, toxic epidermal necrolysis) have been reported. Early symptoms may include flu-like features; if skin or mucosal lesions develop, discontinue ambroxol immediately.
Guaiphenesin
Caution should be exercised in the presence of severe renal or severe hepatic impairment. The concomitant use of cough suppressants is not recommended. Guaiphenesin should not be administered in patients with rare hereditary problems of fructose intolerance. Guaiphenesin is considered to be unsafe in patients with porphyria.
4.5 Drug Interactions
Levosalbutamol
• Other bronchodilators/epinephrine: Use cautiously due to risk of additive cardiovascular effects.
• Beta-blockers: May reduce bronchodilator effect and can precipitate severe bronchospasm in asthma.
• Diuretics (loop/thiazide): May enhance hypokalaemia; use caution, especially with non–potassium-sparing diuretics.
• Digoxin: May reduce serum digoxin levels; monitor levels carefully.
• MAO inhibitors/Tricyclic antidepressants: Use with extreme caution (or within 2 weeks of discontinuation) due to potentiation of cardiovascular effects.
Ambroxol Hydrochloride
Antibiotics: After using ambroxol, the concentrations of antibiotics such as amoxycillin, cefuroxime, erythromycin in bronchial secretions/sputum are increased.
Antitussives: may impair the expectoration of liquefied bronchial mucus due to inhibition of the cough reflex and cause accumulation of secretions.
Guaiphenesin
Paracetamol: Guaiphenesin may increase the rate of absorption of paracetamol.
Laboratory Tests: If urine is collected within 24 hours of a dose of guaiphenesin, its metabolite may cause a color interference with laboratory determinations of urinary 5-hydroxyindoleacetic acid (5-HIAA) and vanillylmandelic acid (VMA).
4.6 Use in Special Population
Pregnancy
There are no adequate, well-controlled studies of this combination in pregnant women. β2-agonists may cause transient neonatal hypoglycaemia. Ambroxol crosses the placenta; while studies (including after 28 weeks) show no harm, its use is not recommended in the first trimester. Guaiphenesin has been associated with a possible increased risk of neural tube defects when used in the first trimester. Therefore, Zorvin-AG Syrup should be avoided in the first trimester and used with caution during the second and third trimesters.
Lactation
It is unknown whether levosalbutamol is excreted in human milk. Ambroxol is excreted in breast milk but is unlikely to harm the infant; data for guaiphenesin are lacking. Use of Zorvin-AG Syrup is not recommended during lactation. A decision should be made to discontinue either breastfeeding or the drug, considering the benefit to the mother.
Paediatric Patients
Safety and efficacy of this formulation in neonates and children below 2 years of age has not been established. Thus, Zorvin-AG Syrup is not recommended for use in paediatric patients below 2 years of age.
Geriatric Patients
Elderly patients with normal renal and hepatic function may be given the same dose as recommended for adults. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Renal Impairment Patients
In severe renal impairment, accumulation of ambroxol metabolites has been reported. Therefore, caution should be exercised while using Zorvin-AG Syrup in patients with significant renal dysfunction. Dose should be reduced or the dosing interval must be extended in patients with severe renal impairment.
Hepatic Impairment Patients
In severe hepatic impairment, Zorvin-AG Syrup should be used with caution.
4.7 Effects on Ability to Drive and Use Machines
Caution should be exercised while taking Zorvin-AG Syrup. If affected by dizziness, palpitation, fine tremors, patients should avoid potentially hazardous tasks such as driving a vehicle or operating machinery.
4.8 Undesirable Effects
Levosalbutamol
The most common adverse effects include palpitations, fine skeletal muscle tremors (especially of the hands), and muscle cramps. Gastrointestinal disturbances such as nausea, vomiting, substernal or epigastric burning, and diarrhoea may also occur. Other reported effects include nervousness, headache, dizziness, fatigue, and insomnia. β2-agonist therapy may cause potentially serious hypokalaemia, which can be worsened by hypoxia. Caution is required in severe asthma, and serum potassium levels should be monitored.
Ambroxol Hydrochloride
Gastrointestinal adverse effects are occasional and usually mild; however, with prolonged high doses, epigastric pain, nausea, and vomiting may occur. Rarely, ambroxol may cause: Gastrointestinal: Dyspepsia, nausea, vomiting, diarrhoea, abdominal pain; Respiratory: Oral/pharyngeal hypoesthesia, dry mouth, dry throat; Nervous system: Dysgeusia (altered taste); Immune system: Anaphylactic reactions, including anaphylactic shock; Skin: Rash, urticaria, pruritus, angioedema, and other hypersensitivity reactions. Allergic reactions such as rash, urticaria, and angioedema may occur in patients hypersensitive to ambroxol.
Guaiphenesin
Adverse effects of guaiphenesin are very rare. Occasionally, high doses may cause gastrointestinal discomfort, nausea, and vomiting. Rare hypersensitivity reactions, including rash, urticaria, angioedema, anaphylaxis, and dyspnoea, have also been reported.
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
4.9 Overdose
Levosalbutamol
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of any of the side effects, e.g. tachycardia, nervousness, headache, tremor, nausea, dizziness, fatigue, and sleeplessness. Hypokalaemia may also occur. As with all sympathomimetic medications, cardiac arrest and even death may be associated with the abuse of levosalbutamol. Treatment consists of discontinuation of levosalbutamol together with appropriate symptomatic therapy. The judicious use of a cardio-selective beta-receptor blocker may be considered, bearing in mind that such medication can produce bronchospasm. There is insufficient evidence to determine if dialysis is beneficial for overdosage of levosalbutamol.
Ambroxol
Acute potential health effects include skin irritation, eye irritation, respiratory tract irritation, gastrointestinal tract irritation with decreased motility or constipation, ulceration or bleeding from the stomach or duodenum, peritonitis. It may even affect behavior/central nervous system (tremor, convulsions, ataxia, and somnolence), respiration (dyspnea, respiratory stimulation), liver, blood (changes if white blood cell count), and urinary system. No data available on chronic potential health effects.
Guaifenesin
The effects of acute toxicity from guaifenesin may include gastrointestinal discomfort, nausea and drowsiness. The drug is, however, rapidly metabolised and excreted in the urine. Patients should be kept under observation and treated symptomatically.
5.0 Pharmacological Properties
5.1 Mechanism of Action
Levosalbutamol
Levosalbutamol is a selective β2-adrenergic agonist that produces bronchodilation by relaxing smooth bronchial muscles. It stimulates β2-receptors, increasing cyclic AMP levels, is associated with the activation of protein kinase A, which in turn, inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in smooth muscle relaxation. It also has mild anti-inflammatory effects by inhibiting the release of bronchoconstrictor and inflammatory mediators from mast cells and eosinophils.
Ambroxol Hydrochloride
Ambroxol causes an increase of secretion in the respiratory tract. It enhances pulmonary surfactant production and stimulates ciliary activity. These actions result in improved mucus flow and transport (mucociliary clearance). Improvement of mucociliary clearance has been shown in clinical pharmacologic studies. Enhancement of fluid secretion and mucociliary clearance facilitates expectoration and reduces cough.
Guaiphenesin
Guaiphenesin is thought to exert its expectorant action by stimulating receptors in the gastric mucosa. This increases the output from secretory glands of the gastrointestinal system and increases the flow of fluids from glands lining the respiratory tract. The result is an increase in volume and decrease in viscosity of bronchial secretions. Another possible mechanism by which it acts is by increasing the water bonding in the sputum, thereby decreasing its viscosity and leading to an increase in mucokinesis. Other actions may include stimulation of vagal nerve endings in bronchial secretory glands and stimulating certain centers in the brain, which in turn enhance respiratory fluid flow.
5.2 Pharmacodynamic Properties
Levosalbutamol
Levosalbutamol, the (R)-enantiomer of salbutamol, is a selective beta 2-receptor agonist. Levosalbutamol has approximately 2-fold greater affinity than racemic salbutamol for the beta 2-adrenergic receptor and approximately 100-fold greater affinity than S-salbutamol. Levosalbutamol is an effective bronchodilator whose primary mechanism of action is unimpeded by (S)-salbutamol. Therefore, when compared with racemic salbutamol, clinically comparable bronchodilation can be achieved with lesser doses of levosalbutamol. Further, levosalbutamol also substantially decreases beta-mediated side effects (tremor, palpitation etc.) associated with racemic salbutamol. Levosalbutamol has bronchodilator, bronchoprotective, anti-inflammatory, and anti-edematous properties.
Ambroxol Hydrochloride
Ambroxol is the active metabolite of bromhexine. Ambroxol is more effective than bromhexine and is non-toxic and well tolerated. Ambroxol possesses mucolytic, mucokinetic (improvement in mucus transport), and secretolytic properties. It promotes the removal of tenacious secretions from the respiratory tract and reduces mucus stasis (arresting the secretion of mucus). Ambroxol also exhibits anti-oxidant activity.
Guaiphenesin
Guaiphenesin produces its expectorant action by increasing the volume of respiratory tract fluid and reducing the viscosity of tenacious secretions.
5.3 Pharmacokinetic Properties
Levosalbutamol: Stability: Stereochemically stable; does not convert to (S)-salbutamol. Absorption: Well absorbed; time to maximum drug concentration (Tmax ) ranges from 45–360 min; oral bioavailability ~9% (single dose) to ~30% (steady state). Distribution: Minimal protein binding; blood/plasma ratio ~1. Metabolism: Mainly intestinal and hepatic sulphate conjugation (SULT1A3); (R)-isomer metabolized faster. Elimination: Primarily renal, with active excretion.
Ambroxol Hydrochloride: Absorption: Rapid and near-complete; bioavailability ~60%; Tmax 0.5–3 hours. Distribution: High protein binding (~90%); widely distributed, highest levels in lungs. Metabolism: Hepatic (mainly CYP3A4-mediated conjugation). Elimination: half-life (t½) ~10 hours; total clearance ~660 mL/min.
Guaiphenesin: Absorption: Well absorbed; Tmax ~15 min. Pharmacokinetics: Cmax ~1.4 mcg/mL (600 mg dose); t½ ~1 hour; not detectable after ~8 hour. Metabolism: Undergoes oxidation and demethylation.
6.0 Nonclinical Properties
6.1 Animal Toxicology or Pharmacology
Non-clinical data revealed no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity, carcinogenic potential, and toxicity to reproduction.
7.0 Description
Each 5 ml of Zorvin-AG Syrup contain 1 mg of levosalbutamol, 30 mg of ambroxol hydrochloride, and 50 mg of guaiphenesin for oral administration.
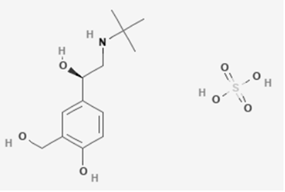
Levosalbutamol
Levosalbutamol, also called as levalbuterol, is a short-acting β2 adrenergic receptor agonist used as a bronchodilator.
Molecular Weight: 337.39 g/mol. Molecular Formula: C13H23NO7S.
Chemical Name: 4-[(1R)-2-(tert-butylamino)-1-hydroxyethyl]-2-(hydroxymethyl)phenol; sulfuric acid.
Ambroxol Hydrochloride
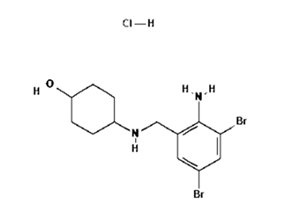
Ambroxol hydrochloride is a metabolite of bromhexine that stimulates mucociliary action and clears the air passages in the respiratory tract.
Molecular Weight: 414.56 g/mol. Molecular Formula: C13H19Br2ClN2O.
Chemical Name: 4-[(2-amino-3,5-dibromophenyl)methylamino]cyclohexan-1 ol; hydrochloride.
Guaiphenesin
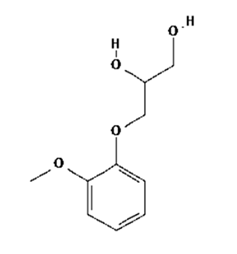
Guaiphenesin, also called as Guaiphenesin or glyceryl guaiacolate, is an expectorant which promotes or facilitates the removal of secretions from the respiratory tract. Guaiphenesin is a white or slightly gray crystalline substance with a slightly bitter aromatic taste.
Molecular Weight: 198.21 g/mol. Molecular Formula: C10H14O4.
Chemical Name: 3-(2-methoxyphenoxy)-1,2-propanediol.
8. Pharmaceutical particulars
8.1 Incompatibilities
None.
8.2 Shelf-Life
Refer on pack.
8.3 Packaging Information
100 ml bottle
8.4 Storage and Handling Instructions
Store below 25°C. Protect from light. Keep out of reach of children.
Do not freeze.
9.0 Patient Counselling Information
- Patients should take Zorvin-AG syrup strictly as prescribed by the physician. Do not exceed the recommended dose or duration of therapy.
- Use during pregnancy: This medicine should be avoided during the first trimester. Use in the second and third trimesters should only be undertaken after consultation with a physician.
- Lactation: Nursing mothers should not use this medicine while breastfeeding unless specifically advised by a healthcare professional.
- Pediatric use: This medicine is not recommended for children below 2 years of age.
- Administration: It is advisable to take this medicine with food to improve tolerability.
- Renal/hepatic impairment: Avoid use in patients with significant renal and/or hepatic dysfunction unless prescribed by a physician.
- Concomitant use: Patients should not use this medicine along with other cough and cold preparations (prescription or over-the-counter) containing similar ingredients without medical advice.
10.0 Date of Revision
16th March 2026



