4.0 Clinical Particulars
4.1 Therapeutic Indications
- For the treatment of mild to moderate hypertension
- For The Management of Hypertension for End-Organ Protection. It Is Reported to Be Useful in Elderly Patients and in Those with Diabetes and Albuminuria. Cilnidipine Has Been Increasingly Used in Patients with Chronic Kidney Disease
4.2 Posology and Method of Administration
In adult patients
Treatment should be individualized on the basis of both effectiveness and tolerance, while not exceeding the maximum recommended daily dose for Cilnidipine.
One tablet once a day administered. Treatment can be initiated with 5-10 mg first, increase to 20 mg daily if necessary.
4.3 Contraindications
Cilnidipine is contraindicated in patients with
- Cardiogenic shock
- Recent history of acute unstable angina or MI
- Severe aortic stenosis
- Hypersensitivity to cilnidipine or any excipient product
- Heart failure
- Hypotension
4.4 Special Warnings and Precautions for use
- Hypotension, poor cardiac reserve, and heart failure.
- Sudden withdrawal may exacerbate angina.
- Discontinue in patients who experience ischemic pain following administration.
4.5 Drug Interactions
Cilnidipine can interact with aldesleukin, quinidine, phenytoin, rifampicin, erythromycin, other anti-hypertensive drugs and anti-psychotic drugs.
4.6 Use in Special Population
Pregnancy
There are no human clinical or animal data concerning the safety of cilnidipine during pregnancy. Until data are available, administration of cilnidipine during pregnancy should be avoided.
Lactation
Nursing mothers should consult a physician before taking Cilnidipine.
Renal impairment
Dose adjustment is not needed in patients with impaired renal function. Cilnidipine at a dose of 10 mg once a day for 7 days in patients with impaired renal function caused no differences in the pharmacokinetic profile compared with that in patients with normal renal function.
Hepatic impairment
Dosage recommendations have not been established in patients with mild to moderate hepatic impairment; therefore, dose selection should be cautious and should start at the lower end of the dosing range. Transient and generally clinically insignificant elevations in SGOT, SGPT, alkaline phosphatase, and serum bilirubin have been reported during calcium antagonist therapy in less than 1% of patients.
4.7 Effects on Ability to Drive and Use Machines
Patients should be advised to avoid operating automobiles and machinery or engaging in other tasks requiring alertness until the patient's response to therapy with cilnidipine tablets has been determined.
4.8 Undesirable Effects
Adverse events which occurred with the use of Cilnidipine are as follows:
- Nervous system: Dizziness, headache, depression, cerebral ischemia
- General: Flushing, peripheral edema, lethargy, tremors, impotence
- Cardiovascular: Hypotension, tachycardia, palpitations, myocardial ischemia
- Gastrointestinal: Gingival hyperplasia, GI disturbances, abnormal liver function
- Special senses: Eye pain
- Other: Myalgia, Transient blindness, Rashes
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
4.9 Overdose
Overdose of cilnidipine may cause excessive reduction in blood pressure. If reduction in blood pressure is remarkable, appropriate measures such as lifting lower extremities, fluid therapy and administration of vasopressors should be taken.
5.0 Pharmacological Properties
5.1 Mechanism of Action
Cilnidipine is a third generation dihydropyridine calcium antagonist with a slow onset and long duration of action. Calcium antagonists inhibit influx of extracellular calcium ions into the cells, resulting in decreased vascular smooth muscle tone and vasodilation, leading to a reduction in blood pressure.
5.2 Pharmacodynamic Properties
Cilnidipine is a dihydropyridine calcium-channel blocker. Dihydropyridine derivative selective calcium-channel blockers with mainly vascular effects. Cilnidipine is a N-type and L-type calcium channel blocker. The effects of cilnidipine on N-type channels give it unique organ-protective properties via the suppression of hyperactivity in the sympathetic nervous system (SNS) and renin-angiotensin-aldosterone system (RAAS). Vasodilatation is caused by Cilnidipine because it inhibits cellular influx of calcium. It has greater selectivity for vascular smooth muscle. It has little or no action at the SA or AV nodes and negative inotropic activity is rarely seen at therapeutic doses.
5.3 Pharmacokinetic Properties
Cilnidipine has been clarified to exert antisympathetic actions in various examinations from cell to human levels, in contrast to classical Ca (2+) channel blockers. After oral administration of cilnidipine it takes 3 hours to reach the peak concentration. Cilnidipine is rapidly metabolized in human liver microsomes and dehydrogenation of dihydropyridine ring of cilnidipine is crucial for the elimination of cilnidipine. Cytochrome P 4503A (CYP3A) is the major human CYP involved in the dehydrogenation of dihydropyridine ring of cilnidipine. After oral administration, large amount of drug could be detected in the gallbladder, bladder, liver and kidney. The α-phase half-life is 1.1 hour.
6.1 Animal Toxicology or Pharmacology
An in vitro study in cultured cells, isolated arteries and cardiac membrane preparations showed that cilnidipine may inhibit vasocontraction by blocking calcium influx via dihydropyridine-sensitive voltage dependent calcium channels, and that the vascular effect of cilnidipine is slower in development and longer in duration compared with nifedipine and nicardipine. In anaesthetised dogs, cilnidipine had a slower onset and longer duration of vasodilator action than nifedipine. Cilnidipine significantly and dose-dependently decreased myocardial oxygen consumption with no significant change in HR. In anaesthetised dogs, cilnidipine as well as nifedipine and nicardipine, caused dose-dependent decreases in arterial BP, and increases in CO, HR and LVdP/dtmax. Intra-arterial administration of cilnidipine resulted in increased flow to several vascular beds, and these vasodilatory effects developed and decayed more slowly than those of nifedipine or nicardipine. In renovascular hypertensive dogs, cilnidipine reduced BP to the same degree as nifedipine or nicardipine but had a significantly longer duration of action. Combinations of cilnidipine with either captopril or atenolol were synergistic. Cilnidipine-induced tachycardia was controlled by atenolol. Cilnidipine reduced BP and increased HR dose-dependently in hypertensive rats. Maximum BP reduction occurred 3h after administration. Although the hypotensive effects of cilnidipine were similar to nifedipine and nicardipine, the onset of action was slower, and the duration of action longer than either of these drugs. Cilnidipine had a weaker negative chronotropic and inotropic effect than nifedipine.
7.0 Description
Zorblok-5/10/20 tablets for oral use contain Cilnidipine 5mg/10mg/20mg.
Cilnidipine is the novel calcium antagonist accompanied with L-type and N-type calcium channel blocking function.
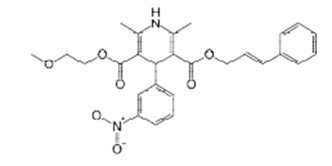
Chemical name: O3-(2-methoxyethyl) O5-[(E)-3-phenylprop-2-enyl] 2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine- 3,5-dicarboxylate.
Molecular formula: C27H28N2O7
Molecular weight: 492.52 g/mol.
8. Pharmaceutical particulars
8.1 Incompatibilities
None.
8.2 Shelf-Life
Refer on pack.
8.3 Packaging Information
As per carton.
8.4 Storage and Handling Instructions
Keep out of reach of children.
Store below 30°C. Protect from light and moisture.
9.0 Patient Counselling Information
- Swallow the whole tablet. Do not crush, chew or break it.
- Try to take it at the same day every day.
- Avoid operating automobiles and machinery or engaging in other tasks requiring alertness until your response to therapy with cilnidipine tablets has been determined.

