4.0 Clinical Particulars
4.1 Therapeutic Indications
- Gastroesophageal Reflux Disease (GERD), erosive reflux esophagitis, prevention of relapse of esophagitis & helps in eradication of H.Pylori associated peptic ulcer.
- Pathological Hypersecretory Conditions Including Zollinger-Ellison Syndrome ulcer.
- Risk Reduction of NSAID Associated Gastric ulcer.
- Prolonged Treatment after IV Induced Prevention of Rebleeding of Peptic ulcer.
4.2 Posology and Method of Administration
Adults
Gastro-Esophageal Reflux Disease (GERD)
- treatment of erosive reflux esophagitis: 40 mg once daily for 4 weeks. An additional 4 weeks treatment is recommended for patients in whom esophagitis has not healed or who have persistent symptoms.
- long-term management of patients with healed esophagitis to prevent relapse: 20 mg once daily.
- symptomatic treatment of gastro-esophageal reflux disease (GERD): 20 mg once daily in patients without esophagitis. If symptom control has not been achieved after 4 weeks, the patient should be further investigated. Once symptoms have resolved, subsequent symptom control can be achieved using 20 mg once daily. An on demand regimen taking 20 mg once daily, when needed, can be used. In NSAID treated patients at risk of developing gastric and duodenal ulcers, subsequent symptom control using an on-demand regimen is not recommended.
In combination with appropriate antibacterial therapeutic regimens for the eradication of Helicobacter pylori and healing of Helicobacter pylori associated duodenal ulcer and prevention of relapse of peptic ulcers in patients with Helicobacter pylori associated ulcers.
20 mg esomeprazole with 1 g amoxicillin and 500 mg clarithromycin, all twice daily for 7 days.
Patients requiring continued NSAID therapy
- healing of gastric ulcers associated with NSAID therapy: The usual dose is 20 mg once daily. The treatment duration is 4-8 weeks.
- prevention of gastric and duodenal ulcers associated with NSAID therapy in patients at risk: 20 mg once daily.
Prolonged treatment after i.v. induced prevention of rebleeding of peptic ulcers
40 mg once daily for 4 weeks after i.v. induced prevention of rebleeding of peptic ulcers.
Treatment of Zollinger Ellison Syndrome
The recommended initial dosage is 40 mg esomeprazole twice daily. The dosage should then be individually adjusted and treatment continued as long as clinically indicated. Based on the clinical data available, the majority of patients can be controlled on doses between 80 to 160 mg esomeprazole daily. With doses above 80 mg daily, the dose should be divided and given twice daily.
Special Populations
Renal impairment: Dose adjustment is not required in patients with impaired renal function. Due to limited experience in patients with severe renal insufficiency, such patients should be treated with caution.
Hepatic impairment: Dose adjustment is not required in patients with mild to moderate liver impairment. For patients with severe liver impairment, a maximum dose of 20 mg esomeprazole should not be exceeded.
Elderly: Dose adjustment is not required in the elderly.
Pediatric population
Adolescents from the age of 12 years
Gastro-Esophageal Reflux Disease (GERD):
- treatment of erosive reflux esophagitis: 40 mg once daily for 4 weeks. An additional 4 weeks treatment is recommended for patients in whom esophagitis has not healed or who have persistent symptoms.
- long-term management of patients with healed esophagitis to prevent relapse: 20 mg once daily.
- symptomatic treatment of gastro-esophageal reflux disease (GERD): 20 mg once daily in patients without esophagitis. If symptom control has not been achieved after 4 weeks, the patient should be further investigated. Once symptoms have resolved, subsequent symptom control can be achieved using 20 mg once daily.
Treatment of duodenal ulcer caused by Helicobacter pylori: When selecting appropriate combination therapy, consideration should be given to official national, regional and local guidance regarding bacterial resistance, duration of treatment (most commonly 7 days but sometimes up to 14 days), and appropriate use of antibacterial agents. The treatment should be supervised by a specialist.
The posology recommendation is:
Weight 30-40 kg: Combination with two antibiotics: Esomeprazole 20 mg, amoxicillin 750 mg and clarithromycin 7.5 mg/kg body weight are all administered together twice daily for one week.
Weight > 40 kg: Combination with two antibiotics: Esomeprazole 20 mg, amoxicillin 1 g and clarithromycin 500 mg all administered together twice daily for one week.
Children below the age of 12 years
Esomeprazole should not be used in children younger than 12 years since no data are available.
Method of administration
The tablets should be swallowed whole with liquid. The tablets should not be chewed or crushed.
4.3 Contraindications
- Hypersensitivity to the active substance, to substituted benzimidazoles or to any of the excipients listed in the formulation.
- Esomeprazole should not be used concomitantly with nelfinavir.
4.4 Special Warnings and Precautions for use
In the presence of any alarm symptom (e.g. significant unintentional weight loss, recurrent vomiting, dysphagia, haematemesis or melaena) and when gastric ulcer is suspected or present, malignancy should be excluded, as treatment with esomeprazole may alleviate symptoms and delay diagnosis.
Long-term use
Patients on long-term treatment (particularly those treated for more than a year) should be kept under regular surveillance.
On-demand treatment
Patients on on-demand treatment should be instructed to contact their physician if their symptoms change in character.
Helicobacter pylori eradication
When prescribing esomeprazole for eradication of Helicobacter pylori, possible drug interactions for all components in the triple therapy should be considered. Clarithromycin is a potent inhibitor of CYP3A4 and hence contraindications and interactions for clarithromycin should be considered when the triple therapy is used in patients concurrently taking other drugs metabolized via CYP3A4 such as cisapride.
Gastrointestinal infections
Treatment with proton pump inhibitors may lead to slightly increased risk of gastrointestinal infections such as Salmonella and Campylobacter.
Absorption of vitamin B12
Esomeprazole, as all acid-blocking medicines, may reduce the absorption of vitamin B12 (cyanocobalamin) due to hypo- or achlorhydria. This should be considered in patients with reduced body stores or risk factors for reduced vitamin B12 absorption on long-term therapy.
Hypomagnesaemia
Severe hypomagnesaemia has been reported in patients treated with proton pump inhibitors (PPIs) like esomeprazole for at least three months, and in most cases for a year. Serious manifestations of hypomagnesaemia such as fatigue, tetany, delirium, convulsions, dizziness and ventricular arrhythmia can occur, but they may begin insidiously and be overlooked. In most affected patients, hypomagnesaemia improved after magnesium replacement and discontinuation of the PPI. For patients expected to be on prolonged treatment or who take PPIs with digoxin or drugs that may cause hypomagnesaemia (e.g., diuretics), health care professionals should consider measuring magnesium levels before starting PPI treatment and periodically during treatment.
Risk of fracture
Proton pump inhibitors, especially if used in high doses and over long durations (>1 year), may modestly increase the risk of hip, wrist and spine fracture, predominantly in the elderly or in presence of other recognized risk factors. Observational studies suggest that proton pump inhibitors may increase the overall risk of fracture by 10–40 %. Some of this increase may be due to other risk factors. Patients at risk of osteoporosis should receive care according to current clinical guidelines and they should have an adequate intake of vitamin D and calcium.
Subacute cutaneous lupus erythematosus (SCLE)
Proton pump inhibitors are associated with very infrequent cases of SCLE. If lesions occur, especially in sun-exposed areas of the skin, and if accompanied by arthralgia, the patient should seek medical help promptly and the health care professional should consider stopping Esomeprazole. SCLE after previous treatment with a proton pump inhibitor may increase the risk of SCLE with other proton pump inhibitors.
Combination with other medicinal products
Co-administration of esomeprazole with atazanavir is not recommended. If the combination of atazanavir with a proton pump inhibitor is judged unavoidable, close clinical monitoring is recommended in combination with an increase in the dose of atazanavir to 400 mg with 100 mg of ritonavir; esomeprazole 20 mg should not be exceeded. Esomeprazole is a CYP2C19 inhibitor. When starting or ending treatment with esomeprazole, the potential for interactions with drugs metabolized through CYP2C19 should be considered. An interaction is observed between clopidogrel and esomeprazole. The clinical relevance of this interaction is uncertain. As a precaution, concomitant use of esomeprazole and clopidogrel should be discouraged.
Severe cutaneous adverse reactions (SCARs)
Severe cutaneous adverse reactions (SCARs) such as erythema multiforme (EM), Stevens Johnson syndrome (SJS), toxic epidermal necrolysis (TEN) and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported very rarely in association with esomeprazole treatment. Patients should be advised of the signs and symptoms of the severe skin reaction EM/SJS/TEN/DRESS and should seek medical advice from their physician immediately when observing any indicative signs or symptoms. Esomeprazole should be discontinued immediately upon signs and symptoms of severe skin reactions and additional medical care/close monitoring should be provided as needed. Re-challenge should not be undertaken in patients with EM/SJS/TEN/DRESS.
Interference with laboratory tests
Increased Chromogranin A (CgA) level may interfere with investigations for neuroendocrine tumors. To avoid this interference, esomeprazole treatment should be stopped for at least 5 days before CgA measurements. If CgA and gastrin levels have not returned to reference range after initial measurement, measurements should be repeated 14 days after cessation of proton pump inhibitor treatment.
4.5 Drug Interactions
Effects of esomeprazole on the pharmacokinetics of other drugs
Protease inhibitors
Omeprazole has been reported to interact with some protease inhibitors. The clinical importance and the mechanisms behind these reported interactions are not always known. Increased gastric pH during omeprazole treatment may change the absorption of the protease inhibitors. Other possible interaction mechanisms are via inhibition of CYP2C19. For atazanavir and nelfinavir, decreased serum levels have been reported when given together with omeprazole and concomitant administration is not recommended. Co administration of omeprazole (40 mg once daily) with atazanavir 300 mg/ritonavir 100 mg to healthy volunteers resulted in a substantial reduction in atazanavir exposure (approximately 75 % decrease in AUC, Cmax and Cmin). Increasing the atazanavir dose to 400 mg did not compensate for the impact of omeprazole on atazanavir exposure. The co administration of omeprazole (20 mg qd) with atazanavir 400 mg/ritonavir 100 mg to healthy volunteers resulted in a decrease of approximately 30 % in the atazanavir exposure as compared with the exposure observed with atazanavir 300 mg/ritonavir 100 mg qd without omeprazole 20 mg qd. Co-administration of omeprazole (40 mg qd) reduced mean nelfinavir AUC, Cmax and Cmin by 36–39 % and mean AUC, Cmax and Cmin for the pharmacologically active metabolite M8 was reduced by 75-92 %. Due to the similar pharmacodynamic effects and pharmacokinetic properties of omeprazole and esomeprazole, concomitant administration with esomeprazole and atazanavir is not recommended and concomitant administration with esomeprazole and nelfinavir is contraindicated. For saquinavir (with concomitant ritonavir), increased serum levels (80-100 %) have been reported during concomitant omeprazole treatment (40 mg qd). Treatment with omeprazole 20 mg qd had no effect on the exposure of darunavir (with concomitant ritonavir) and amprenavir (with concomitant ritonavir). Treatment with esomeprazole 20 mg qd had no effect on the exposure of amprenavir (with and without concomitant ritonavir). Treatment with omeprazole 40 mg qd had no effect on the exposure of lopinavir (with concomitant ritonavir).
Methotrexate
When given together with PPIs, methotrexate levels have been reported to increase in some patients. In high-dose methotrexate administration a temporary withdrawal of esomeprazole may need to be considered.
Tacrolimus
Concomitant administration of esomeprazole has been reported to increase the serum levels of tacrolimus. A reinforced monitoring of tacrolimus concentrations as well as renal function (creatinine clearance) should be performed, and dosage of tacrolimus adjusted if needed.
Medicinal products with pH dependent absorption
Gastric acid suppression during treatment with esomeprazole and other PPIs might decrease or increase the absorption of medicinal products with a gastric pH dependent absorption. As with other medicinal products that decrease intragastric acidity, the absorption of medicinal products such as ketoconazole, itraconazole and erlotinib can decrease and the absorption of digoxin can increase during treatment with esomeprazole. Concomitant treatment with omeprazole (20 mg daily) and digoxin in healthy subjects increased the bioavailability of digoxin by 10 % (up to 30 % in two out of ten subjects). Digoxin toxicity has been rarely reported. However, caution should be exercised when esomeprazole is given at high doses in elderly patients. Therapeutic drug monitoring of digoxin should then be reinforced.
Medicinal products metabolized by CYP2C19
Esomeprazole inhibits CYP2C19, the major esomeprazole metabolizing enzyme. Thus, when esomeprazole is combined with drugs metabolized by CYP2C19, such as diazepam, citalopram, imipramine, clomipramine, phenytoin, etc., the plasma concentrations of these drugs may be increased, and a dose reduction could be needed. This should be considered especially when prescribing esomeprazole for on-demand therapy.
Diazepam
Concomitant administration of 30 mg esomeprazole resulted in a 45 % decrease in clearance of the CYP2C19 substrate diazepam.
Phenytoin
Concomitant administration of 40 mg esomeprazole resulted in a 13 % increase in trough plasma levels of phenytoin in epileptic patients. It is recommended to monitor the plasma concentrations of phenytoin when treatment with esomeprazole is introduced or withdrawn.
Voriconazole
Omeprazole (40 mg once daily) increased voriconazole (a CYP2C19 substrate) Cmax and AUCτ by 15 % and 41 %, respectively.
Cilostazol
Omeprazole as well as esomeprazole act as inhibitors of CYP2C19. Omeprazole, given in doses of 40 mg to healthy subjects in a cross-over study, increased Cmax and AUC for cilostazol by 18 % and 26 % respectively, and one of its active metabolites by 29 % and 69 % respectively.
Cisapride
In healthy volunteers, concomitant administration of 40 mg esomeprazole resulted in a 32 % increase in area under the plasma concentration-time curve (AUC) and a 31 % prolongation of elimination half-life (t1/2) but no significant increase in peak plasma levels of cisapride. The slightly prolonged QTc interval observed after administration of cisapride alone, was not further prolonged when cisapride was given in combination with esomeprazole.
Warfarin
Concomitant administration of 40 mg esomeprazole to warfarin-treated patients in a clinical trial showed that coagulation times were within the accepted range. However, post marketing, a few isolated cases of elevated INR of clinical significance have been reported during concomitant treatment. Monitoring is recommended when initiating and ending concomitant esomeprazole treatment during treatment with warfarin or other coumarine derivatives.
Clopidogrel
Results from studies in healthy subjects have shown a pharmacokinetic (PK)/pharmacodynamic (PD) interaction between clopidogrel (300 mg loading dose/75 mg daily maintenance dose) and esomeprazole (40 mg p.o. daily) resulting in decreased exposure to the active metabolite of clopidogrel by an average of 40% and resulting in decreased maximum inhibition of (ADP induced) platelet aggregation by an average of 14 %. When clopidogrel was given together with a fixed dose combination of esomeprazole 20 mg + ASA 81 mg compared to clopidogrel alone in a study in healthy subjects there was a decreased exposure by almost 40 % of the active metabolite of clopidogrel. However, the maximum levels of inhibition of (ADP induced) platelet aggregation in these subjects were the same in the clopidogrel and the clopidogrel + the combined (esomeprazole + ASA) product groups. Inconsistent data on the clinical implications of a PK/PD interaction of esomeprazole in terms of major cardiovascular events have been reported from both observational and clinical studies. As a precaution concomitant use of clopidogrel should be discouraged.
Investigated medicinal products with no clinically relevant interaction
Amoxicillin and quinidine: Esomeprazole has been shown to have no clinically relevant effects on the pharmacokinetics of amoxicillin or quinidine.
Naproxen or rofecoxib: Studies evaluating concomitant administration of esomeprazole and either naproxen or rofecoxib did not identify any clinically relevant pharmacokinetic interactions during short term studies.
Effects of other medicinal products on the pharmacokinetics of esomeprazole
Medicinal products which inhibit CYP2C19 and/or CYP3A4: Esomeprazole is metabolised by CYP2C19 and CYP3A4. Concomitant administration of esomeprazole and a CYP3A4 inhibitor, clarithromycin (500 mg b.i.d.), resulted in a doubling of the exposure (AUC) to esomeprazole. Concomitant administration of esomeprazole and a combined inhibitor of CYP2C19 and CYP 3A4 may result in more than doubling of the esomeprazole exposure. The CYP2C19 and CYP3A4 inhibitor voriconazole increased omeprazole AUCτ by 280%. A dose adjustment of esomeprazole is not regularly required in either of these situations. However, dose adjustment should be considered in patients with severe hepatic impairment and if long-term treatment is indicated.
Medicinal products which induce CYP2C19 and/or CYP3A4: Drugs known to induce CYP2C19 or CYP3A4 or both (such as rifampicin and St. John's wort) may lead to decreased esomeprazole serum levels by increasing the esomeprazole metabolism.
Paediatric population
Interaction studies have only been performed in adults.
4.6 Use in Special Population
Pregnancy
Clinical data on exposed pregnancies with esomeprazole are insufficient. With the racemic mixture, omeprazole data on a larger number of exposed pregnancies from epidemiological studies indicate no malformative nor foetotoxic effect. Animal studies with esomeprazole do not indicate direct or indirect harmful effects with respect to embryonal/foetal development. Animal studies with the racemic mixture do not indicate direct or indirect harmful effects with respect to pregnancy, parturition or postnatal development. Caution should be exercised when prescribing to pregnant women. A moderate amount of data on pregnant women (between 300-1000 pregnancy outcomes) indicates no malformative or foeto/neonatal toxicity of esomeprazole. Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity.
Breastfeeding
It is not known whether esomeprazole is excreted in human breast milk. There is insufficient information on the effects of esomeprazole in newborns/infants. Esomeprazole should not be used during breastfeeding.
Fertility
Animal studies with the racemic mixture omeprazole, given by oral administration do not indicate effects with respect to fertility.
4.7 Effects on Ability to Drive and Use Machines
Esomeprazole has minor influence on the ability to drive and use machines. Adverse reactions such as dizziness (uncommon) and blurred vision (rare) has been reported. If affected patients should not drive or use machines.
4.8 Undesirable Effects
| System Organ Class | Frequency | Undesirable Effect |
|---|
| blood and lymphatic system disorders | rare | leukopenia, thrombocytopenia |
| very rare | agranulocytosis, pancytopenia |
| immune system disorders | rare | hypersensitivity reactions e.g. fever, angioedema and anaphylactic reaction/shock |
| metabolism and nutrition disorders | uncommon | peripheral oedema |
| rare | hyponatraemia |
| not known | hypomagnesaemia; Severe hypomagnesaemia can correlate with hypocalcaemia. Hypomagnesaemia may also be associated with hypokalaemia. |
| psychiatric disorders | uncommon | insomnia |
| rare | agitation, confusion, depression |
| very rare | aggression, hallucinations |
| nervous system disorders | common | headache |
| uncommon | dizziness, paraesthesia, somnolence |
| rare | taste disturbance |
| eye disorders | rare | blurred vision |
| ear and labyrinth disorders | uncommon | vertigo |
| respiratory, thoracic and mediastinal disorders | rare | bronchospasm |
| gastrointestinal disorders | common | abdominal pain, constipation, diarrhoea, flatulence, nausea/vomiting, fundic gland polyps (benign) |
| uncommon | dry mouth |
| rare | stomatitis, gastrointestinal candidiasis |
| not known | microscopic colitis |
| hepatobiliary disorders | uncommon | increased liver enzymes |
| rare | hepatitis with or without jaundice |
| very rare | hepatic failure, encephalopathy in patients with pre-existing liver disease |
| skin and subcutaneous tissue disorders | uncommon | dermatitis, pruritus, rash, urticaria |
| rare | alopecia, photosensitivity |
| very rare | erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS) |
| not known | Subacute cutaneous lupus erythematosus. |
| musculoskeletal and connective tissue disorders | uncommon | fracture of the hip, wrist or spine |
| rare | arthralgia, myalgia |
| very rare | muscular weakness |
| renal and urinary disorders | very rare | interstitial nephritis; in some patients renal failure has been reported concomitantly. |
| reproductive system and breast disorders | very rare | gynaecomastia |
| general disorders and administration site conditions | rare | malaise, increased sweating |
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
4.9 Overdose
There is very limited experience to date with deliberate overdose. The symptoms described in connection with 280 mg were gastrointestinal symptoms and weakness. Single doses of 80 mg esomeprazole were uneventful. No specific antidote is known. Esomeprazole is extensively plasma protein bound and is therefore not readily dialyzable. As in any case of overdose, treatment should be symptomatic and general supportive measures should be utilised.
5.0 Pharmacological Properties
5.1 Mechanism of Action
Esomeprazole is the S-isomer of omeprazole and reduces gastric acid secretion through a specific targeted mechanism of action. It is a specific inhibitor of the acid pump in the parietal cell. Both the R- and S-isomer of omeprazole have similar pharmacodynamic activity.
5.2 Pharmacodynamic Properties
Esomeprazole is a weak base and is concentrated and converted to the active form in the highly acidic environment of the secretory canaliculi of the parietal cell, where it inhibits the enzyme HK-ATPase – the acid pump and inhibits both basal and stimulated acid secretion.
Pharmacodynamic effects
After oral dosing with esomeprazole 20 mg and 40 mg the onset of effect occurs within one hour. After repeated administration with 20 mg esomeprazole once daily for five days, mean peak acid output after pentagastrin stimulation is decreased 90 % when measured 6-7 hours after dosing on day five. After five days of oral dosing with 20 mg and 40 mg of esomeprazole, intragastric pH above 4 was maintained for a mean time of 13 hours and 17 hours, respectively over 24 hours in symptomatic GERD patients. The proportion of patients maintaining an intragastric pH above 4 for at least 8, 12 and 16 hours respectively were for esomeprazole 20 mg 76 %, 54 % and 24 %. Corresponding proportions for esomeprazole 40 mg were 97 %, 92 % and 56 %. Using AUC as a surrogate parameter for plasma concentration, a relationship between inhibition of acid secretion and exposure has been shown. Healing of reflux esophagitis with esomeprazole 40 mg occurs in approximately 78 % of patients after four weeks, and in 93 % after eight weeks. One week treatment with esomeprazole 20 mg b.i.d. and appropriate antibiotics, results in successful eradication of H. pylori in approximately 90 % of patients. After eradication treatment for one week there is no need for subsequent monotherapy with antisecretory drugs for effective ulcer healing and symptom resolution in uncomplicated duodenal ulcers. In a randomized, double blind, placebo-controlled clinical study, patients with endoscopically confirmed peptic ulcer bleeding characterized as Forrest Ia, Ib, IIa or IIb (9 %, 43 %, 38 % and 10 % respectively) were randomized to receive esomeprazole solution for infusion (n=375) or placebo (n=389). Following endoscopic haemostasis, patients received either 80 mg esomeprazole as an intravenous infusion over 30 minutes followed by a continuous infusion of 8 mg per hour or placebo for 72 hours. After the initial 72-hour period, all patients received open label 40 mg oral esomeprazole for 27 days for acid suppression. The occurrence of rebleeding within 3 days was 5.9 % in the esomeprazole treated group compared to 10.3 % for the placebo group. At 30 days post-treatment, the occurrence of rebleeding in the esomeprazole treated versus the placebo treated group was 7.7 % vs 13.6 %. During treatment with antisecretory medicinal products, serum gastrin increases in response to the decreased acid secretion. Also, CgA increases due to decreased gastric acidity. The increased CgA level may interfere with investigations for neuroendocrine tumours. Available published evidence suggests that proton pump inhibitors should be discontinued between 5 days and 2 weeks prior to CgA measurements. This is to allow CgA levels that might be spuriously elevated following PPI treatment to return to reference range. An increased number of ECL cells, possibly related to the increased serum gastrin levels, have been observed in both children and adults during long-term treatment with esomeprazole. The findings are considered to be of no clinical significance. During long-term treatment with antisecretory drugs gastric glandular cysts have been reported to occur at a somewhat increased frequency. These changes are a physiological consequence of pronounced inhibition of acid secretion, are benign and appear to be reversible. Decreased gastric acidity due to any means including proton pump inhibitors, increases gastric counts of bacteria normally present in the gastrointestinal tract. Treatment with proton pump inhibitors may lead to slightly increased risk of gastrointestinal infections such as Salmonella and Campylobacter and, in hospitalized patients, possibly also Clostridium difficile.
5.3 Pharmacokinetic Properties
Absorption
Esomeprazole is acid labile and is administered orally as gastro-resistant pellets. In vivo conversion to the R-isomer is negligible. Absorption of esomeprazole is rapid, with peak plasma levels occurring approximately 1-2 hours after dose. The absolute bioavailability is 64 % after a single dose of 40 mg and increases to 89 % after repeated once-daily administration. For 20 mg esomeprazole the corresponding values are 50 % and 68 %, respectively. Food intake both delays and decreases the absorption of esomeprazole although this has no significant influence on the effect of esomeprazole on intragastric acidity.
Distribution
The apparent volume of distribution at steady state in healthy subjects is approximately 0.22 l/kg body weight. Esomeprazole is 97 % plasma protein bound.
Biotransformation
Esomeprazole is completely metabolized by the cytochrome P450 system (CYP). The major part of the metabolism of esomeprazole is dependent on the polymorphic CYP2C19, responsible for the formation of the hydroxy- and desmethyl metabolites of esomeprazole. The remaining part is dependent on another specific isoform, CYP3A4, responsible for the formation of esomeprazole sulphone, the main metabolite in plasma.
Elimination
The parameters below reflect mainly the pharmacokinetics in individuals with a functional CYP2C19 enzyme, extensive metabolizers. Total plasma clearance is about 17 l/h after a single dose and about 9 l/h after repeated administration. The plasma elimination half-life is about 1.3 hours after repeated once-daily dosing. Esomeprazole is completely eliminated from plasma between doses with no tendency for accumulation during once-daily administration. The major metabolites of esomeprazole have no effect on gastric acid secretion. Almost 80 % of an oral dose of esomeprazole is excreted as metabolites in the urine, the remainder in the faeces. Less than 1 % of the parent drug is found in urine.
6.1 Animal Toxicology or Pharmacology
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity, carcinogenic potential, toxicity to reproduction and development. Adverse reactions not observed in clinical studies but seen in animals at exposure levels similar to clinical exposure levels and with possible relevance to clinical use were as follows: Carcinogenicity studies in the rat with the racemic mixture have shown gastric ECL-cell hyperplasia and carcinoids. These gastric effects in the rat are the result of sustained, pronounced hypergastrinemia secondary to reduced production of gastric acid and are observed after long-term treatment in the rat with inhibitors of gastric acid secretion.
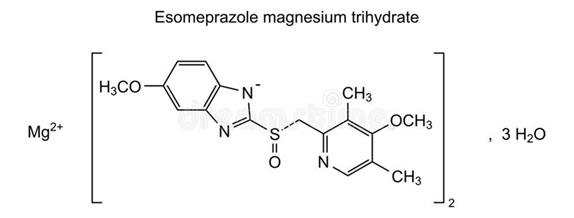
7.0 Description
Esomeprazole is the S-isomer of omeprazole, which is a mixture of the S- and R isomers.
Chemical Name: magnesium;bis(5-methoxy-2-[(S)-(4-methoxy-3,5-dimethyl-2-pyridinyl)methylsulfinyl]benzimidazol-1-ide);trihydrate
Molecular formula: C34H42MgN6O9S2
Molecular weight: 767.2 g/mol
8. Pharmaceutical particulars
8.1 Incompatibilities
Not applicable
8.2 Shelf-Life
Refer on pack
8.3 Packaging Information
10 Tablets Strip
8.4 Storage and Handling Instructions
Store protected from light & moisture, at a temperature not exceeding 30°C.
9.0 Patient Counselling Information
- Acute Tubulointerstitial Nephritis: Advise the patient or caregiver to call the patient's healthcare provider immediately if they experience signs and/or symptoms associated with suspected acute TIN.
- Clostridium difficile-Associated Diarrhea: Advise the patient or caregiver to immediately call the patient's healthcare provider if they experience diarrhea that does not improve.
- Bone Fracture: Advise the patient or caregiver to report any fractures, especially of the hip, wrist or spine, to the patient's healthcare provider.
- Severe Cutaneous Adverse Reactions: Advise the patient or caregiver to discontinue Esomeprazole and immediately call the patient's healthcare provider for at first appearance of a severe cutaneous adverse reaction or other sign of hypersensitivity signs or symptoms associated with Severe Cutaneous Adverse Reactions.
- Cutaneous and Systemic Lupus Erythematosus: Advise the patient or caregiver to immediately call the patient's healthcare provider for any new or worsening of symptoms associated with cutaneous or systemic lupus erythematosus.
- Cyanocobalamin (Vitamin B-12) Deficiency: Advise the patient or caregiver to report any clinical symptoms that may be associated with cyanocobalamin deficiency to the patient's healthcare provider if they have been receiving Esomeprazole for longer than 3 years.
- Hypomagnesemia and Mineral Metabolism: Advise the patient or caregiver to report any clinical symptoms that may be associated with hypomagnesemia, hypocalcemia, and/or hypokalemia to the patient's healthcare provider, if they have been receiving Esomeprazole for at least 3 months.
- Drug Interactions: Advise the patient or caregiver to report to their healthcare provider if starting treatment with rilpivirine-containing products, clopidogrel, St. John's Wort or rifampin; or, if they take high-dose methotrexate.

