1.0 Generic Name
Levodropropizine syrup
2.0 Qualitative and Quantitative Composition
Each 5 ml contains:
Levodropropizine IP……………………………….30 mg
3.0 Dosage Form and Strength
Syrup 30 mg/5 ml


Levodropropizine syrup
Each 5 ml contains:
Levodropropizine IP……………………………….30 mg
Syrup 30 mg/5 ml
Symptomatic treatment of non-productive cough.
Adults and children from 12 years and older
Maximum 60 mg levodropropizine (10 ml syrup), three times daily. The time between administrations should be at least 6 hours.
Children over 2 years
1 mg/kg bodyweight given three times daily for a total daily doses of 3mg/kg body weight at intervals of at least 6 hours.
Levodropropizine syrup contraindicated in children below 2 years.
Elderly
In elderly patients, levodropropizine is used with caution; a risk of changed pharmacokinetics linked to age can be present.
Renal and Hepatic Impairment
In case of severe renal (creatin clearance below 35ml/min) or in case of severe hepatic failure, the benefit-risk ratio should be taken into consideration.
Method of administration: Preferably, the syrup will be taken away from meals with an interval of at least 6 hours between administrations. The period of treatment should remain brief; treatment should be discontinued as soon as the symptoms have disappeared.
Pregnancy
There are not enough clinical data studying the use of levodropropizine during pregnancy to evaluate the potential toxicity. Tusvia syrup is contraindicated during pregnancy.
Lactation
In animal studies it was demonstrated that levodropropizine is excreted in maternal milk. Therefore, Tusvia syrup is contra-indicated during breast feeding.
Fertility
There are no data available.
In exceptional cases, somnolence and vertigo have been reported. Caution is advised when driving or operating a machine.
| System Organ Class | Common (≥1/100 to <1/10) | Very Rare (<1/10,000) |
|---|---|---|
| Cardiac disorders | Palpitations | Tachycardia |
| Gastro-intestinal disorders | Nausea, pyrosis, dyspepsia, diarrhea, vomiting | Abdominal pain, stomach pain |
| Skin and subcutaneous disorders | Cutaneous allergic reactions | Urticaria, erythema, exanthema, pruritis, angio-edema |
| Nervous system disorders | — | Tremor, paresthesia |
| Respiratory, thoracic and mediastinal disorders | — | Dyspnea, cough, bronchial edema |
| Musculoskeletal and connective tissue disorders | — | Weakness of the internal membranes |
| Vascular disorders | — | Hypotension |
| General disorders | — | Discomfort, feeling of weakness |
| Immune system disorders | — | Anaphylactic reaction |
| Psychiatric disorders | — | Depersonalisation |
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
Significant undesirable effects have not been observed following ingestion of a single dose up to 240 mg or following ingestion of multiple 120 mg doses, 3 times daily for 8 consecutive days. In case of an overdose, a temporary transitional tachycardia can be expected. General measures for treating a drug overdose apply (gastric lavage, activated charcoal, giving parenteral fluid).
Levodropropizine inhibit the cough reflex by acting on the peripheral receptors and their afferent conductors. Levodropropizine inhibits the C-fibers of the vagus nerves and modulate the sensory neuropeptides production in the respiratory tract, involved in the cough reflex. Levodropropizine has a dose-dependent and short-term local anaesthetic activity. It also has a mild analgesic and an antihistaminic action.
Levodropropizine is a peripherally acting antitussive working at tracheobronchial level. The peripheral action has been demonstrated in animal studies. Its mechanism provides this drug antitussive properties against cough associated to different lung pathologies, but without relevant central side effects. Levodropropizine inhibits bronchospasms induced by histamine, serotonin and bradykinin. Levodropropizine exerts its antitussive effect through an inhibitory action at the level of the airway sensory nerves involving modulation of sensitive C-fibers and release of neuropeptides.
Absorption
Bioavailability of levodropropizine was found to be greater than 75% after oral administration. Plasma protein binding rate was lower (11-14%).
Distribution
In human, oral levodropropizine was rapidly absorbed and distributed throughout the body.
Biotransformation
There is no data about the specific site of metabolism of levodropropizine either in the liver or in other sites.
Elimination
Plasma elimination half-life of levodropropizine is approximately 1-2 hours. Its excretion is mainly in the urine. Elimination of the active substance is either in the form of both unchanged and conjugated or free levodropropizine or in conjugated p-hydroxy-levodropropizine metabolites. Elimination of the active substance and its metabolites in 48 hours approximates to 35% of the administered dose. Results of the repeat dose studies have demonstrated that 8 days of treatment (3 times a day) did not alter the elimination characteristics of the drug and therefore accumulation or metabolic auto-induction were unlikely.
Acute oral toxicity is 886.5 mg/kg, 1287mg/kg and 2492 mg/kg, respectively in rats, mice and guinea pigs. In guinea pigs, the therapeutic index calculated as the ratio of LD50/ED50after oral administration was 16 to 53, depending on the cough induction model. Toxicity tests for repeated oral administrations have shown that the daily dose without toxic effect corresponds to 24 mg/kg.
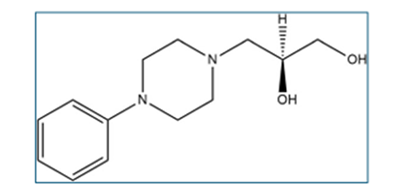
Levodropropizine belongs to class of medicines called antitussives. Its chemical name is (2S)-3-(4-Phenylpiperazin-1-yl)propane-1,2-diol.
Molecular formula: C13H20N2O2
Molecular weight: 236.315 g/mol
Not applicable
Refer on pack
100 ml Bottle
Store at a temperature not exceeding 30°C. Protect from light.