4.4 Special Warnings and Precautions for use
Pancreatitis
Acute pancreatitis, including fatal pancreatitis, has been reported in patients treated with linagliptin. Take careful notice of potential signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue linagliptin and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using linagliptin.
Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
Insulin secretagogues and insulin are known to cause hypoglycemia. The risk of hypoglycemia is increased when linagliptin is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. The use of linagliptin in combination with insulin in subjects with severe renal impairment was associated with a higher rate of hypoglycemia. Therefore, a lower dosage of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with linagliptin.
Hypersensitivity Reactions
There have been reports of serious hypersensitivity reactions in patients treated with linagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions occurred predominantly within the first 3 months after initiation of treatment with linagliptin, with some reports occurring after the first dose. If a serious hypersensitivity reaction is suspected, discontinue linagliptin, assess for other potential causes for the event, and institute alternative treatment for diabetes mellitus. Angioedema has also been reported with other dipeptidyl peptidase-4 (DPP-4) inhibitors. Use caution in a patient with a history of angioedema to another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with linagliptin.
Severe and Disabling Arthralgia
There have been reports of severe and disabling arthralgia in patients taking linagliptin. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider the drug as a possible cause for severe joint pain and discontinue drug if appropriate.
Bullous Pemphigoid
Bullous pemphigoid was reported in 7 (0.2%) patients treated with linagliptin compared to none in patients treated with placebo and 3 of these patients were hospitalized due to bullous pemphigoid. Cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving linagliptin. If bullous pemphigoid is suspected, linagliptin should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
Heart Failure
An association between DPP-4 inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease. Consider the risks and benefits of linagliptin prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of linagliptin.
4.5 Drug Interactions
Inducers of P-glycoprotein or CYP3A4 Enzymes
Rifampin decreased linagliptin exposure, suggesting that the efficacy of Linectra-5 may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. Therefore, use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer
Insulin Secretagogues or Insulin
Insulin and insulin secretagogues are known to cause hypoglycemia. The risk of hypoglycemia is increased when linagliptin is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. Coadministration of Linectra-5 with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia.
4.6 Use in Special Population
Pregnancy
The use of linagliptin has not been studied in pregnant women. Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity. As a precautionary measure, it is preferable to avoid the use of linagliptin during pregnancy.
Breast-feeding
Available pharmacokinetic data in animals have shown excretion of linagliptin/metabolites in milk. A risk to the breast-fed child cannot be excluded. A decision must be made whether to discontinue breast-feeding or to discontinue/abstain from linagliptin therapy considering the benefit of breast-feeding for the child and the benefit of therapy for the woman.
Fertility
No studies on the effect on human fertility have been conducted for linagliptin. Animal studies do not indicate direct or indirect harmful effects with respect to fertility.
4.7 Effects on Ability to Drive and Use Machines
Linagliptin has no or negligible influence on the ability to drive and use machines. However, patients should be alerted to the risk of hypoglycaemia especially when combined with sulphonylurea and/or insulin
4.8 Undesirable Effects
Table 1:Tabulated list of adverse reactions
|
System organ class
Adverse reaction
|
Frequency of adverse reaction
|
|
Infections and infestations
|
|
|
Nasopharyngitis
|
uncommon
|
|
Immune system disorders
|
|
|
Hypersensitivity (e.g. bronchial hyperreactivity)
|
uncommon
|
|
Metabolism and nutrition disorders
|
|
|
Hypoglycemia
|
very common
|
|
Respiratory, thoracic and mediastinal disorders
|
|
|
Cough
|
uncommon
|
|
Gastrointestinal disorders
|
|
|
Pancreatitis
|
rare
|
|
Constipation
|
uncommon
|
|
Skin and subcutaneous tissue disorders
|
|
|
Angioedema
|
rare
|
|
Urticaria
|
rare
|
|
Rash
|
uncommon
|
|
Bullous pemphigoid
|
rare
|
|
Investigations
|
|
|
Amylase increased
|
uncommon
|
|
Lipase increased
|
common
|
Reporting of Suspected Adverse Reactions
Reporting suspected adverse reactions after authorization of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via email to: medico@zorvia.com
By reporting side effects, you can help provide more information on the safety of this medicine.
4.9 Overdose
Symptoms
During controlled clinical trials in healthy subjects, single doses of up to 600 mg linagliptin (equivalent to 120 times the recommended dose) were generally well tolerated. There is no experience with doses above 600 mg in humans.
Therapy
In the event of an overdose, it is reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring and institute clinical measures if required.
5.0 Pharmacological Properties
5.1 Mechanism of Action
Linagliptin is an inhibitor of DPP-4, an enzyme that degrades the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Thus, linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin in a glucose-dependent manner and decreasing the levels of glucagon in the circulation. Both incretin hormones are involved in the physiological regulation of glucose homeostasis. Incretin hormones are secreted at a low basal level throughout the day and levels rise immediately after meal intake. GLP-1 and GIP increase insulin biosynthesis and secretion from pancreatic beta cells in the presence of normal and elevated blood glucose levels. Furthermore, GLP-1 also reduces glucagon secretion from pancreatic alpha-cells, resulting in a reduction in hepatic glucose output.
5.2 Pharmacodynamic Properties
Linagliptin binds to DPP-4 in a reversible manner and thus increases the concentrations of incretin hormones. Linagliptin glucose-dependently increases insulin secretion and lowers glucagon secretion, thus resulting in better regulation of glucose homeostasis. Linagliptin binds selectively to DPP-4 and selectively inhibits DPP 4, but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, 4-way crossover study, 36 healthy subjects were administered a single oral dose of linagliptin 5 mg, linagliptin 100 mg (20 times the recommended dose), moxifloxacin, and placebo. No increase in QTc was observed with either the recommended dose of 5 mg or the 100 mg dose. At the 100 mg dose, peak linagliptin plasma concentrations were approximately 38-fold higher than the peak concentrations following a 5 mg dose.
5.3 Pharmacokinetic Properties
Absorption
The absolute bioavailability of linagliptin is approximately 30%. A high-fat meal reduced Cmax by 15% and increased AUC by 4%; this effect is not clinically relevant. Linectra-5 may be administered with or without food.
Distribution
The mean apparent volume of distribution at steady-state following a single intravenous dose of linagliptin 5 mg to healthy subjects is approximately 1,110 L, indicating that linagliptin extensively distributes to the tissues. Plasma protein binding of linagliptin is concentration-dependent, decreasing from about 99% at 1 nmol/L to 75%-89% at ≥30 nmol/L, reflecting saturation of binding to DPP-4 with increasing concentration of linagliptin. At high concentrations, where DPP-4 is fully saturated, 70% to 80% of linagliptin remains bound to plasma proteins and 20% to 30% is unbound in plasma. Plasma binding is not altered in patients with renal or hepatic impairment.
Elimination
Linagliptin has a terminal half-life of about 200 hours at steady state, though the accumulation half-life is about 11 hours. Renal clearance at steady state was approximately 70 mL/min.
Metabolism
Following oral administration, the majority (about 90%) of linagliptin is excreted unchanged, indicating that metabolism represents a minor elimination pathway. A small fraction of absorbed linagliptin is metabolized to a pharmacologically inactive metabolite, which shows a steady-state exposure of 13.3% relative to linagliptin.
Excretion
Following administration of an oral [14C]-linagliptin dose to healthy subjects, approximately 85% of the administered radioactivity was eliminated via the enterohepatic system (80%) or urine (5%) within 4 days of dosing.
6.0 Nonclinical Properties
6.1 Animal Toxicology or Pharmacology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Linagliptin did not increase the incidence of tumors in male and female rats in a 2-year study at doses of 6, 18, and 60 mg/kg. The highest dose of 60 mg/kg is approximately 418 times the clinical dose of 5 mg/day based on AUC exposure. Linagliptin did not increase the incidence of tumors in mice in a 2-year study at doses up to 80 mg/kg (males) and 25 mg/kg (females), or approximately 35- and 270-times the clinical dose based on AUC exposure. Higher doses of linagliptin in female mice (80 mg/kg) increased the incidence of lymphoma at approximately 215-times the clinical dose based on AUC exposure. Linagliptin was not mutagenic or clastogenic with or without metabolic activation in the Ames bacterial mutagenicity assay, a chromosomal aberration test in human lymphocytes, and an in vivo micronucleus assay. In fertility studies in rats, linagliptin had no adverse effects on early embryonic development, mating, fertility, or bearing live young up to the highest dose of 240 mg/kg (approximately 943-times the clinical dose based on AUC exposure).
7.0 Description
Linectra-5 tablets for oral use contain linagliptin, an inhibitor of the DPP-4 enzyme.
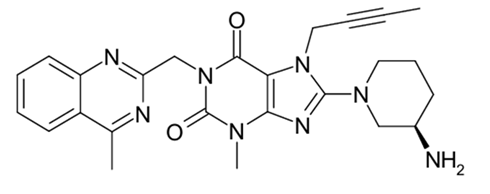
Chemical name: 1H-Purine-2,6-dione, 8-[(3R)-3-amino-1-piperidinyl]-7-(2-butyn-1-yl)-3,7-dihydro-3-methyl-1-[(4-methyl-2-quinazolinyl)methyl]-
Molecular formula: C25H28N8O2
Molecular weight: 472.54 g/mol.
8.0 Pharmaceutical Particulars
8.1 Incompatibilities
None.
8.2 Shelf-Life
Refer on pack.
8.3 Packaging Information
As per carton.
8.4 Storage and Handling Instructions
Keep out of reach of children.
Store below 30°C in a dry place. Protect from light.
9.0 Patient Counselling Information
Pancreatitis
Inform patients that acute pancreatitis has been reported during use of Linectra-5. Inform patients that persistent severe abdominal pain, sometimes radiating to the back, which may or may not be accompanied by vomiting, is the hallmark symptom of acute pancreatitis. Instruct patients to discontinue Linectra-5 promptly and contact their healthcare provider if persistent severe abdominal pain occurs.
Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
Inform patients that the incidence of hypoglycemia is increased when Linectra-5 is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin.
Hypersensitivity Reactions
Inform patients that serious allergic reactions, such as anaphylaxis, angioedema, and exfoliative skin conditions, have been reported with use of Linectra-5. If symptoms of allergic reactions (such as rash, skin flaking or peeling, urticaria, swelling of the skin, or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing) occur, patients must stop taking Linectra-5 and seek medical advice promptly.
Severe and Disabling Arthralgia
Inform patients that severe and disabling joint pain may occur with this class of drugs. The time to onset of symptoms can range from one day to years. Instruct patients to seek medical advice if severe joint pain occurs.
Bullous Pemphigoid
Inform patients that bullous pemphigoids have been reported during use of Linectra-5. Instruct patients to seek medical advice if blisters or erosion occur.
Heart Failure
Inform patients of the signs and symptoms of heart failure. Before initiating Linectra-5, patients should be asked about a history of heart failure or other risk factors for heart failure including moderate to severe renal impairment. Instruct patients to contact their healthcare provider as soon as possible if they experience symptoms of heart failure, including increasing shortness of breath, rapid increase in weight or swelling of the feet.
Missed Dose
Instruct patients to take Linectra-5 only as prescribed. If a dose is missed, it should be taken as soon as the patient remembers. Advise patients not to double their next dose.

